Chemicals ProductsBromine Group
Calcium Bromide
Calcium bromide solutions are used in the oil industry as auxiliaries for oil drillings. These solutions, which are pumped in the ground do not of course have to be as pure as those used for production of medicines, foodstuffs, air conditioners, etc.
Nevertheless, to satisfy market requirements regarding environmental protection and prevention of soil and water pollution, it is necessary to purify these solutions. It is important to note that we benefit twice from using byproducts. First because we save on production costs by using cheaper raw materials obtained from byproducts. Second, because we save on the treatment of acid solutions which, if not used, would be hazardous waste requiring neutralization.
For many years calcium bromide solutions have been prepared by an acid-base reaction between an aqueous solution of hydrogen bromide and solid calcium hydroxide, which is also called slaked lime.
Sodium Bromide
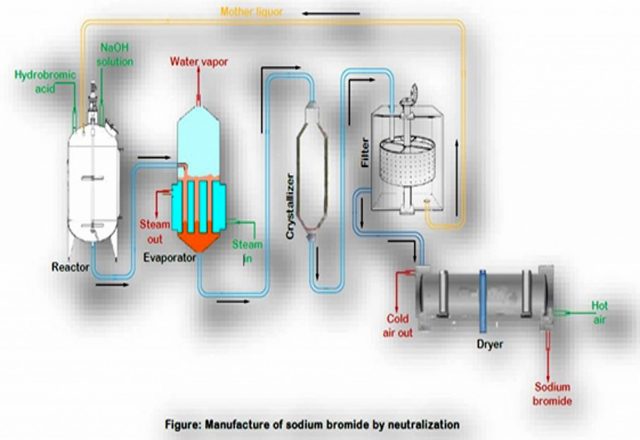
Sodium bromide (NaBr) is an inorganic compound. It is a high-melting white, crystalline solid that resembles sodium chloride. It is a widely used source of the bromide ion and has many applications. Sodium bromide is the most available alkali bromide and salt of hydrobromic acid. It is available in the form of crystals or powder. From aqueous solution sodium bromide crystallizes as a dihydrate and NaBr2.H2O at below 510C, while above it crystallizes as the anhydrous compound. NaBr crystallizes in the same cubic motif as NaCl, NaF and NaI. The anhydrous salt crystallizes above 50.70C. It is produced by treating sodium hydroxide with hydrogen bromide.
Sodium bromide is the most useful inorganic bromide in industry.[7] It is also used as a catalyst in TEMPO-mediated oxidation reactions.
Disinfectant:
Sodium bromide is used in conjunction with chlorine as a disinfectant for hot tubs and swimming pools
Petroleum Industry:
Sodium bromide is used to prepare dense fluids used in oil wells.